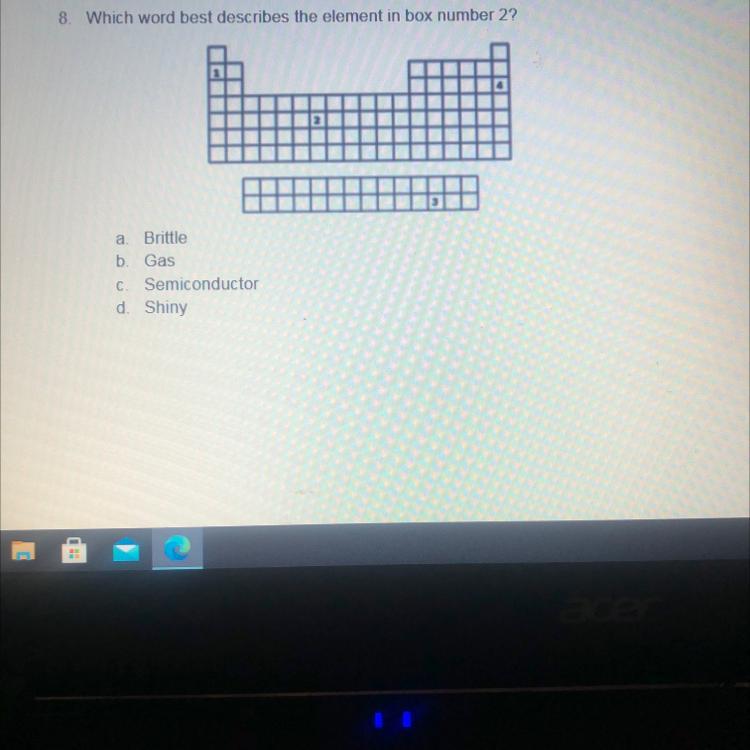
8. Which word best describes the element in box number 2?
a а Brittle
b Gas
С Semiconductor
d. Shiny
Answers
Answer:
gas
Explanation:
i think it might be gas
Related Questions
What is the specific heat of a metal if the temperature of a 12.5 g sample increases from 19.5 C to 33.6 C when it absorbs 37.7 J of heat.
Answers
Answer:
0.213 J/g°C
Explanation:
To calculate specific heat of the metal, the formula is used:
Q = m × c × ∆T
Where Q = amount of heat
m = mass
c = specific heat
∆T = change in temperature
According to this question, Q = 37.7 J, m= 12.5 g, initial temperature= 19.5 °C, final temperature = 33.6°C, c=?
Q = m × c × ∆T
37.7 = 12.5 × c × (33.6-19.5)
37.7 = 12.5c × 14.1
37.7 = 176.25c
c = 37.7/176.25
c = 0.2139
Hence, the specific heat of the metal is 0.213 J/g°C
Destructive forces can include floods, landslides, tornadoes, tsunamis, wildfires and
volcanoes.
True
False
Answers
Answer:
answer is true
Answer:
True
Explanation:
please help.
In 1976, an airplane was flown at a speed of 2193 miles per hour. How many kilometers could the plane fly in 4.3 hours? Use D.A. with these conversion factors: 1mile = 5280ft 1ft =12 inches 1 inch = 2.54cm
Answers
Answer:
9429.9
Explanation:
periodt
what is atomic radius
Answers
Explanation:
The atomic radius of a chemical element is a measure of the size of its atoms, usually the mean or typical distance from the center of the nucleus to the boundary of the surrounding shells of electrons. Since the boundary is not a well-defined physical entity, there are various non-equivalent definitions of atomic radius. Three widely used definitions of atomic radius are: Van der Waals radius, ionic radius, and covalent radius
True or False: A Precipitate is a solid
that forms out of a liquid solution as a
result of a chemical reaction
Answers
Answer:
true because it undergoes a tranformation
Explanation:
A piece of metal that has a density of 5.2 g/cm3 and a mass of 100 g was placed in a full jar of water. How many mL of water was spilled out of the jar? Note: one cm^3 = one mL
Answers
Answer:
100g / (5.2g/cm3)
= 100g / (5.2g / 1cm3)
= 100g x 1cm3 / 5.2 g
= 19.2 cm3
Since 1 cm3 = 1 ml, your answer is 19.2 ml.
The given density and mass of the metallic piece can be used to estimate the volume of the substance. The metal with a mass of 100 gm and a density of 5.2 g/cm³ has a volume of 19.23 ml.
What is the relation between density and volume?The density of the substance has been given by the ratio of mass to the volume of the substance in g/cm³ or g/mL. It shows the packaging of the molecules in the substances and has been in inverse relation to the volume.
The mass being constant and an increase in volume decreases the density of the substance and vice versa. The volume can be calculated as:
Volume = mass ÷ density
Given,
Density of the metal (ρ) = 5.2g/cm³
Mass of the metal piece (m) = 100g
Volume = V
Substituting the values above as:
ρ = m ÷ V
V = m ÷ ρ
V = 100 ÷ 5.2
= 19.23 mL
Therefore, 19.23 mL is the volume of water that spilled out of the jar.
Learn more about volume and density, here:
https://brainly.com/question/13799762
#SPJ2
Insoluble substances can dissolve in all solvents
True or false
Answers
Answer:
Not all substances dissolve in all solvents
Explanation:
In chemistry, a common rule for determining if a solvent will dissolve a given solute is "like dissolves like." Solvents composed of polar molecules, such as water, dissolve other polar molecules, such as table salt, while nonpolar solvents, such as gasoline, dissolve nonpolar substances such as wax. Some substances are strong enough to not dissolve.
After lab, all of Darrel's friends looked at his data and laughed and laughed. They told him that he was 30.8% too low in the boiling point he had just recorded. He had recorded a boiling point of 50o C on his data sheet. What is the correct boiling point of the liquid he was working with in lab?
Answers
Answer:
100°
Explanation:
For a boiling point is it always 100°
Predict how James will improve the view of the sample. How will the magnification and resolution of the image change?
Answers
Answer:
answer questions brain
What is the mass of an object with a density of 5.7 g/cm^3 and a volume of 48
cm^3? *
Answers
Answer:
273.6 g
Explanation:
mass =(density)(volume)
mass=(5.7g/cm^3 )(48 cm^3)
cancel out your cm^3 and your are left with grams
multiply (5.7g)(48)=273.6 g
The mass of an object with a density of 5.7 g/cm³ and a volume of 48 cm³ is 273.6 grams.
What is density?Density is defined as the degree to which a material is packed together.
It is also defined as a mass per unit volume of the substance.
Its SI unit is kilogram per cubic meter or gram per meter.
Density can be expressed as
Density = mass / volume
Mass = density x volume
As given, Density = 5.7 g/cm³
Volume = 48 cm³
So, Mass = 5.7 g/cm³ x 48 cm³
= 273.6 grams
Thus, the mass of an object with a density of 5.7 g/cm³ and a volume of 48 cm³ is 273.6 grams.
To learn more about density, refer to the link below:
https://brainly.com/question/15164682
#SPJ5
describe how the current modern atomic theory and model differs from the model jj Thompson proposed ?
Answers
A helium-filled balloon is released into the atmosphere. As the balloon rises, which
would MOST likely increase and cause the balloon to burst?
O the volume of the helium
O the mass of the helium
o the temperature of the helium
O the density of the helium
Answers
what is the solution to the equation below rounded to the correct number of significant figures? 4700 L - 281.4 = _____ L
Answers
Answer:4400
I went over this problem multiple times and I finally found the soultion
The concept of significant figures are mainly used by scientist and engineer to know the significance of digits in a measurement. Therefore, 4400L is the solution to the equation rounded to the correct number of significant figures.
What is significant figures?Significant figures are the figures that indicate the degree of accuracy of a value. It tells about the precision of a value. It gives an idea about the digits that are necessary to indicate the experimental value.
Rules for counting significant figures are:
Number between 1 to 9 is always significant
Zeroes after a number has got no significance
Zeroes before a number has got no significance
Zeroes between number has got significance
4700 L - 281.4 =4400L
Therefore, 4400L is the solution to the equation rounded to the correct number of significant figures.
To learn more about Significant figures, here:
https://brainly.com/question/12656148?
#SPJ2
What happens if you mix lithium and aluminum together?
Answers
Answer:
hello
Explanation:
do you meant something like that
please hurry i’m taking a test
Answers
Answer:
im pretty sure its move at a constant rate to the right
How do covalent bonds form?
Answers
10 POINTS
Which tool can be used to measure the volume of a liquid to one decimal place?
Options:
a beaker
a graduated cylinder
an Erlenmeyer flask
a test tube
Answers
a graduated cylinder
how does litmus indicator work
Answers
The substance which responds to the change in the concentration of the hydronium ions in solution by changing its color is known as an indicator.
What is true about the formation of a bond?
Answers
Answer:
There is an overall release of energy when bonds form.
Explanation:
There is a general release of energy when bonds form. This energy is called bond energy.
Bond energy is involved in the breakdown or formation of one or more bonds between atoms of a molecule. Atoms bond with each other to achieve their electronic stability, that is, they move from a higher energy situation to a lower energy one. With this we can state that when the bond between atoms is formed, energy is released; therefore, its breakdown depends on energy absorption.
How would the particles of a solid, liquid, and gas ALL at room temperature compare? *
A.solid particles have lower average kinetic energy
B.the gas particles are larger than both solid and liquid
C.the pas particles are moving the slowest
D.the spacing of the particles is different, but all have the same average kinetic energy
Plz help
Answers
Answer:
A
Explanation:
Solid particles only vibrate, so they have very little kinetic energy.
For B, the size of the particles stay the same, just phase/state changes, so not B.
For C, gas particles move the fastest compared to liquid and solid given the same environment.
For D, they don't have the same kinetic energy because solid particles barely move while gas particles keep moving.
name two diffirent kinds of animals and identify at least one characteristic that makes them diffirent from each other
Answers
Answer:
Bears and Whales
Explanation:
Bears keep themselves warm via a thick fur coat
Whales keep themselves warm with a thick layer of blubber
These models show the electron structures of two different nonmetal elements. Element 1 at left has a purple circle at center with 2 concentric black lines around it. The first line has 2 small green balls on it. The second line has 8 small green balls on it. Element 2 at right has a purple center with 5 concentric circles around it, with the first circle innermost. The first circle has 2 small green balls on it, and the second circle has 8 small green balls on it. The third circle has 18 small green balls on it, and the fourth circle has 18 small green balls on it. The fifth circle has 6 small green balls on it. Which element is likely more reactive, and why?
Answers
Answer:
Element 2
Explanation:
If we look at the model stated for element 1, it is clear that element 1 must be a noble gas. It has eight electrons in its outermost shell this implies that it has already attained a complete octet of electrons and is reluctant towards chemical reaction.
The second element belongs to group 16 since it has six electrons on its outermost shell. It is certainly more reactive than element 1 which is a noble gas.
Answer: D
Element 2 is more reactive because it does not have a full valence shell, so it will attract electrons.
Explanation: Answer on Edgenuity
please help:)!!!!
important
Answers
Answer:
I beleive its the middle/second option :)
Explanation:
Sorry if I'm wrong but Newton's third law is "for every action (force) in nature there is an equal and opposite reaction." meaning if object A exerts a force on object B, then object B also exerts an equal and opposite force on object A so I beleive the moon would push up on the astronaut with a force of 300 N
I hope this helps, have a good day :)
The modern synthesis combined the concepts of _______ and evolution.
Answers
Molecular Evidence.
Which is the IUPAC name for NO?
nitric oxide
binitrogen oxide
dinitrogen dioxide
nitrogen monoxide
Answers
The IUPAC name for NO is nitric oxide. The correct option is A.
What is IUPAC naming?The International Union of Pure and Applied Chemistry (IUPAC) is a global organization that represents chemistry as well as related sciences and technologies.
The chemical compound's identity is represented by its common and IUPAC names, which differ from one another. Every chemical compound has multiple names.
Complete the following step-by-step response: The IUPAC nomenclature is a standardized name given to organic compounds in accordance with official naming rules.
Nitric oxide, also known as NO, is a colorless gas. It is one of the most important nitrogen oxides.
Nitric oxide is a free radical, which means it has an unpaired electron, which is sometimes represented in its chemical formula by a dot.
Thus, the correct option is A.
For more details regarding IUPAC, visit:
https://brainly.com/question/16631447
#SPJ6
Answer:
D: Nitrogen Monoxide
Explanation:
There's one Nitrogen atom and one Oxygen atom
Which quantum number describes the orientation of an orbital within a sublevel?
Answers
Explanation:
Each orbital within a particular sublevel is distinguished by its value of ml. This quantum number may be more aptly named the orbital orientation quantum number. In each energy sublevel (designated by l) there are 2l+1 possible independent orientations of the electron cloud.
Answer:
[tex]m_l-magnetic[/tex]
Explanation:
The principal energy level, n, tells us which shell it is in and the total energy of the electron.
The angular momentum (azimuthal), l, tells us which sublevel/subshells the electron is in.
The magnetic, m sub l, tells us the orientation the electron is spinning.
Please help ASAP 100 points points help I will mark you as brainlister
Answers
this is lithium ion with a +1 charge
What is the formula for an alkene containing six carbon atoms?
A. C6H10
B. C2H6
C. C6H12
D. C5H12
Answers
Answer:
C. C₆H₁₂
Explanation:
Alkenes are the unsaturated hydrocarbons which form a homologous series with a general formula;
Cₙ H₂ₙ
n in this problem is 6
2n = 2 x 6 = 12
The formula of the compound will be C₆H₁₂
Which properties change the composition of a substance?
A chemical properties
B physical properties
C neither chemical nor physical properties
D chemical and physical properties
Answers
Answer:
D. Chemical and physical properties
By what process do small molecules move into cells?
Answers
Answer:
small molecules move into the cell, Then it is by diffusion or facilliated diffusion.